New Drug Would possibly Scale back Sickle Cell Ache Crises
 By Steven Reinberg
By Steven ReinbergHealthDay Reporter
SATURDAY, Dec. three, 2016 (HealthDay Information) -- An experimental drug could assist cut back episodes of ache related to sickle cell illness, a brand new examine finds.
Outcomes of an early trial confirmed the drug -- known as SelG1 for now -- decreased episodes of sickle cell-related ache crises by 45 p.c. As well as, the drug appeared protected and was well-tolerated, researchers say.
"Sufferers with sickle cell illness have issues, the commonest of which is ache crises that require medical care and generally hospitalization," mentioned lead researcher Dr. Kenneth Ataga. He is director of the sickle cell program on the College of North Carolina at Chapel Hill.
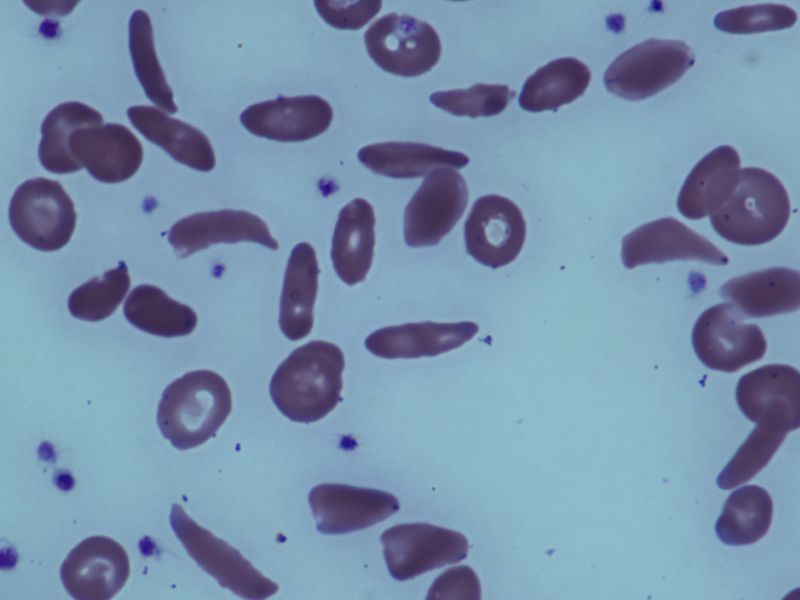
Folks with inherited sickle cell illness have abnormally formed purple blood cells. These sickle-shaped cells persist with the partitions of blood vessels, slowing and blocking regular blood movement. Blockage ends in much less blood and oxygen reaching cells, which in flip causes extreme muscle ache.
In line with the U.S. Facilities for Illness Management and Prevention, roughly 100,000 People have sickle cell illness, with blacks disproportionately affected.
"We do not have many remedies for this ache," Ataga mentioned. At present, just one drug, hydroxyurea, is accessible to assist forestall these episodes. And plenty of sufferers proceed to have acute ache episodes regardless of the remedy.
SelG1, an antibody, works otherwise from hydroxyurea, Ataga mentioned. SelG1 assaults the molecule P-selectin, which is accountable for blocking regular blood movement. By stopping P-selectin, the drug retains blood flowing, thus stopping the ache crises, he defined.
For his or her examine, Ataga and colleagues randomly assigned 198 sufferers with sickle cell illness to obtain one in all two IV doses of SelG1 or a placebo. Sufferers acquired an preliminary dose, one other dose two weeks later, then a dose each 4 weeks for 50 weeks for a complete of 14 doses.
Over the yr of the examine, sufferers who acquired the very best dose of SelG1 skilled a 45 p.c discount within the fee of sickle cell ache crises, the researchers discovered.
Unwanted effects occurred in 5 p.c or extra of sufferers receiving SelG1. These included joint ache, itching, vomiting, chest ache, diarrhea, fatigue, muscle ache and abdomen ache, the examine reported.
The trial was the second of three analysis phases required for drug approval in america.
Since this trial, the drug large Novartis has acquired Selexys Prescription drugs Corp., which developed SelG1 and funded the examine. Novartis has not determined if or when it's going to begin bigger trials, an organization spokeswoman mentioned.
"It will be good to have multiple drug to supply folks," mentioned Dr. Maggie Fader, a pediatric hematologist oncologist at Nicklaus Kids's Hospital in Miami. "Nonetheless, there is not sufficient info but to say that this drug goes to be higher than what we've," she mentioned.
Additionally, the examine lasted solely 12 months, so how properly sufferers reply over time is not identified, Fader mentioned. Bigger and longer research are wanted, she added.
Sufferers who are suffering from sickle cell ache crises generally strive dangerous remedies or use highly effective opioid painkillers, which may end up in dependence and the necessity for more and more greater doses, Fader mentioned.
"The bulk are going to have important ache crises the place they require narcotics or hospitalizations," she added. "One thing that is a disease-modifying agent which may cut back these issues could be welcome."
The examine outcomes had been scheduled for presentation Sunday on the assembly of the American Society of Hematology, in San Diego. They will even be printed Dec. three within the New England Journal of Drugs.

Copyright © 2016 HealthDay. All rights reserved.
SOURCES: Kenneth Ataga, M.B.B.S, professor, medication, director, Complete Sickle Cell Program, College of North Carolina, Chapel Hill; Maggie Fader, M.D., pediatric hematologist oncologist, Nicklaus Kids's Hospital, Miami; Dec. three, 2016, New England Journal of Drugs; Dec. four, 2016, presentation, American Society of Hematology assembly, San Diego
No comments:
Post a Comment